Study unravels key molecule role in Sars-CoV-2 replication
26/05/2022
Maíra Menezes (IOC/Fiocruz)
In a study recently published in the scientific journal Nature, one of the most important in the world, researchers from the Oswaldo Cruz Foundation (Fiocruz) and the Telethon Institute of Genetics and Medicine (Tigem) in Italy, uncover the role of a key molecule for the replication of the Sars-CoV-2 virus, the agent of COVID-19. In an unprecedented way, scientists characterize the performance of nonstructural protein 6 (known by the acronym NSP6) in the formation of viral replication sites inside the host cell. The research also shows that several variants of coronavirus present increased activity of this molecule due to a genetic variation, which can help the virus to replicate more efficiently within the cell.
"Most studies on coronavirus variants focus on mutations that alter the surface of the viral particle, which can help the virus escape the action of antibodies and facilitate entry into the cell, where it can replicate and, establish the infection. In our work, we look at another aspect: the replicative capacity of the virus. We identified that a change in the NSP6 protein, which occurs in different genetic variants of concern, represents a gain in function for the pathogen," says the head of the Immunopharmacology Laboratory of the Oswaldo Cruz Institute (IOC/Fiocruz) and one of the authors of the article, Patricia Bozza.
The research characterized the performance of nonstructural protein 6 (known by the acronym NSP6) in the formation of viral replication sites inside the host cell (image: Gutemberg Brito, IOC/Fiocruz)
At least six coronavirus variants of concern have a set of deletions in the genome – that is, the absence of snippets of genetic code – which results in the production of a small-sized NSP6 protein. Among these are two strains that had widespread: alpha, which emerged in the United Kingdom at the end of 2020 and spread rapidly in the country, and gamma, which emerged in Brazil in the same period and was responsible for the largest number of pandemic deaths in the country.
In addition, in the BA.2 strain of the omicron variant, the gene encoding the NSP6 protein also presents variations, suggesting that there is selective pressure on the function of this protein. According to the World Health Organization (WHO), this strain is currently predominant in more than 60 countries.
Mutations in the virus genome occur randomly, but those that confer some advantage tend to be selected positively and can spread more, generating a greater number of cases. "Changes in the NSP6 protein make virus replication more efficient within the cell. By characterizing the action of this protein and deepening knowledge about the replication process of Sars-CoV-2, our study provides new bases for the development of direct-action therapies against coronavirus," explains the researcher at the Center for Technological Development in Health (CDTS) and one of the authors of the study, Thiago Moreno Lopes e Souza.
Replicative organelle
Viruses do not have the ability to multiply on their own. Therefore, they need to invade a cell and hijack the cellular machinery for replication of its genetic material and production of viral proteins. At the same time, they must protect themselves from the cell's defense mechanisms, which try to prevent this attack.
It was already known that coronavirus is capable of creating special organelles within the cell for its replication. These organelles are double-membrane envelope vesicles, inside which the production of copies of the genetic material of the virus occurs in a protected way.
However, the mechanisms involved in the formation of these organelles were poorly explored and no previous study had detailed the function of the NSP6 protein in this process. Using various analysis techniques from cell biology, biotechnology and virology, the scientists discovered that this molecule performs three essential functions for the assembly and functioning of replicative organelles.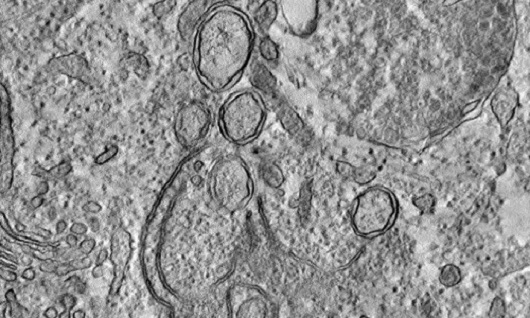
The NSP6 protein forms thin connecting channels and acts as a filter between the SARS-CoV-2 replicative organelles and the endoplasmic reticulum (image: Ricciardi et al; Nature)
First, the NSP6 protein forms thin connecting channels and acts as a filter between the SARS-CoV-2 replicative organelles and the endoplasmic reticulum – an important cellular organelle, composed of a tubes and vesicles network where the production of several molecules occurs. “We observed that it allows the entry of lipids [fatty molecules] into the replicative vesicle, but blocks most of the proteins, which could be the cell's weapons against the coronavirus”, points out Patrícia.
NSP6's performance is also important to position and organizing replicative organelles, forming structures similar to grape clusters. In the trials, the researchers observed that, in the absence of this protein, the number of replicative organelles in each cluster decreased from 15 to three and the shape of the organelles became irregular.
In addition, the protein promotes the association between replicative vesicles and lipid corpuscles – lipid-rich organelles present inside the cell. "NSP6 attracts lipid corpuscles close to replicative organelles. This is a way for the virus to drain the metabolic pathways of the cell for its replication. In addition to being an important source of energy, lipids are necessary to form the membranes of the replicative vesicles and for the synthesis of new viral particles, because the virus has a lipid-rich envelope", says Patrícia.
The alteration of the NSP6 protein identified in several variants of coronavirus makes the molecule more active. The researchers noted that, compared to the virus that initially emerged in Wuhan, China, the variants have more replicative organelles, larger and faster in size, in addition to the high presence of the NSP6 molecule in connections to the endoplasmic reticulum.
"The deletions in the genome also make NSP6 more hydrophobic, with a better ability to attract lipid corpuscles, bringing them closer to replicative compartments and manipulating the cell's energy flow," says Thiago.
Cooperation in search of answers
The NSP6 functions characterization opens doors for the development and improvement of therapies. “When we understand the molecular mechanism, we can look for drugs that act on these processes. Considering that viral genome replication occurs within lipid membranes, we can also consider that lipophilic substances [which are attracted to lipids] can increase the antivirals action that act by inhibiting RNA synthesis”, comments Thiago.
The study was coordinated by Tigem and had the collaboration of the University of Naples Federico II and the Experimental Zooprophylactic Institute of Southern Italy, in addition to the IOC and CDTS from Fiocruz. Graduate students from the Cellular and Molecular Biology Program at the IOC and the Immunology Program at the Federal University of Rio de Janeiro (UFRJ) also participated in the work.
The partnership between Fiocruz researchers and Italian scientists is an example of the quickly establishing scientific collaborations' importance in dealing with the pandemic. The research groups had never worked together, but they identified common interests and complementary research abilities through manuscripts released in pre-print format, which anticipate the research results dissemination, before the peer review process that precedes the article's publication in scientific journals.
In Italy, the researchers worked with mechanisms of trafficking in cell membranes and the plasmids construction, genetic structures that are used to induce cells to produce specific virus proteins. At Fiocruz, scientists were already developing studies on the importance of lipid metabolism in viral replication and on cell infection with variants of concern for Sars-CoV-2, a type of work that can only be carried out in laboratories with a biosafety level 3. The joint efforts allowed the researchers to develop a complete study that contributes to elucidating the fundamental question about how the virus manages to take over cellular structures for its replication.
"The pandemic has accelerated the process of scientific cooperation, because accelerated dissemination of results facilitates collaboration between researchers who already have projects in progress," points out Thiago. "Our work is the result of an international effort to respond to the pandemic. As a result, we achieved more robust results, faster", emphasizes Patrícia.





