Fiocruz processes 40% of RT-PCR tests of Brazil’s public health system
28/06/2021
Ricardo Valverde (Agência Fiocruz de Notícias)
Over the past week, the four central units of large processing of molecular tests (RT-PCR) for Covid-19 coordinated by the Ministry of Health, with the support of Fiocruz, achieved the equivalent of 7 million samples processed, which represents about 40% of all testing done in the public system. In addition to processing the tests, Fiocruz is also responsible for their production, having already concluded its committment, signed in 2020 with the Ministry of Health (MS), of delivering 11.7 million molecular tests for Sars-CoV-2. The tests are still being delivered according to the Ministry’s demand and schedule, attending to the strategy of testing and distribution.
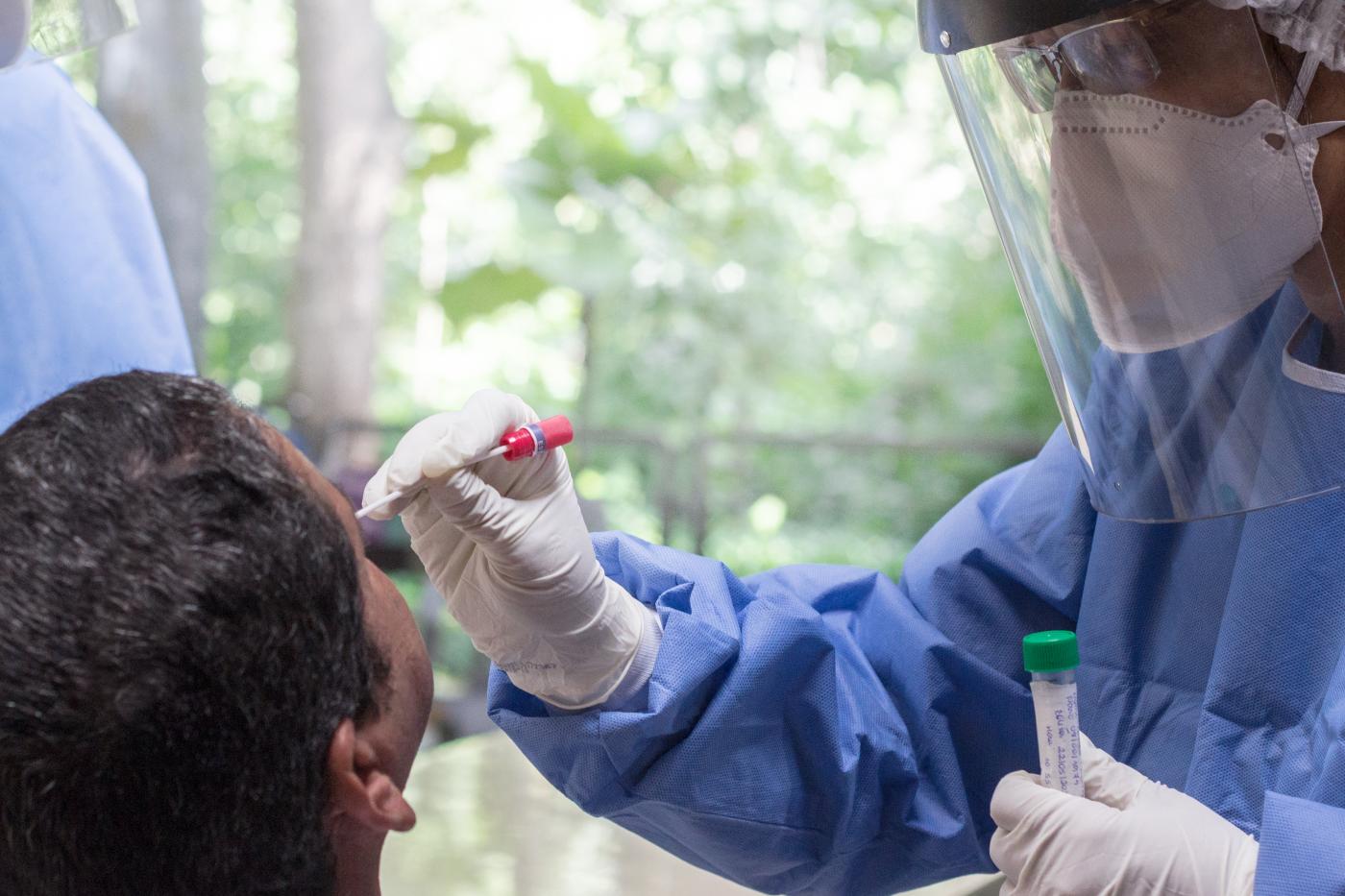 The four central units built to support the expansion of testing in the surveillance network have an operational capacity of up to 280 thousand samples per week, with an average usage of 70%, and have received samples from 23 states, catering to 48 institutions (Lacen - Central Laboratories, municipal laboratories, city governments, regional entities etc) until now. The operation is non-stop, 24 hours a day, 7 days a week.
The four central units built to support the expansion of testing in the surveillance network have an operational capacity of up to 280 thousand samples per week, with an average usage of 70%, and have received samples from 23 states, catering to 48 institutions (Lacen - Central Laboratories, municipal laboratories, city governments, regional entities etc) until now. The operation is non-stop, 24 hours a day, 7 days a week.
For the leading executive of the Support Office for the Operational Management of RT-PCR Testing Chain, Maria Clara Lippi, “the large-scale operation of the central units makes it possible to process a high volume of samples an an extremely fast and accurate manner. The operational capacity of all other central units allows for a quick response to demands, especially when it comes to absorbing peaks of testing, which require a dynamic response, without relevant impact on the average processing time.
Of the four large processing central units coordinated by the Ministry of Health in a partnershiph with Fiocruz, two are directly administrated by the Foundation, through the Vice-Presidency of Production and Innovation in Health. Under the name of Support to Covid-19 Diagnosis (Unadig), one is being installed at the Manguinhos campus, in Rio de Janeiro, and the other in the unit located in the state of Ceará. The other two, in São Paulo and Paraná, are the result of partnerships with the Emergency Diagnosis Center (Dasa Network) and the Paraná Institute of Molecular Biology (IBMP), respectively. The operations began in early April (Dasa and IBMP), July (Rio de Janeiro), and August (Ceará) 2020.
Management and technology guarantee agile sample results
According to Camila Guindalini, coordinator of Fiocruz’s Support Office for the Operational Management of RT-PCR Testing Chain, the central units combine “automated platforms with high-tech equipment that make it possible to expand the large-scale processing of samples from patients suspected to have Covid-19”.
The office’s leading executive, Maria Clara, mentions that all sample processing phases, since they are received by Fiocruz, can be followed online. “This is a virtual tour that displays the entire process, from receiving the sample to analyzing the result”. The communication system between the different processing rooms was designed based on principles of biosafety and of boosting the different steps. In addition to the rapidity of the process, the system minimizes risks and preserves the integrity of the material.
The work begins with a manual operation of verification and organization. “This receiving phase prepares and standardizes the samples to be processed in the automated flow of analysis. The samples then go through an extraction process in which the genetic material is separated, as it is the specific focus of the analysis. The material is then submitted to the RT-PCR step, in which it will be amplified and later analyzed, verifying whether the sample is positive or negative for Sars-CoV-2, detectable or not detectable. The average processing time of the samples since they are received by one of the four centra processing units until the result is released is 17 hours”, explains Camila, the coordinator.
Logistics and transportation to the states
Fiocruz is responsible for transporting the collection kits and other critical consumables for the molecular testing chaiin to the states, as well as the samples to be processed, picking them up from the laboratories and delivering them to the central units, with a level of service of up to 24 hours.
The most recent consolidated figures, up to the end of May, show that 3,828 transportation of biological sample operations were carried out, for a total of 99 tons transported and 14.4 million collection kits.


